Many traditional technologies for the production of industrial chemicals are highly energy-intensive owing to the extreme conditions required, such as high temperature and pressure. Ammonia for example, a key component in fertilizers, is typically produced using the Haber-Bosch process, at temperatures of over 400°C and pressures up to 100 atm.
The production of ammonia consumes 5% of global energy production and accounts for 1% of all global greenhouse gas emissions.
What’s the alternative?
An alternative to such energy-intensive traditional processes are electrochemical reactions that utilize a catalyst for targeted synthesis of chemicals. Such electrochemical reactions often operate at room temperature and pressure, and can have the potential to significantly reduce the carbon footprint of industrial chemical synthesis. However, current electrochemical technologies are limited by the low efficiency of known catalyst materials.
Discovering new high-performing catalysts is crucial.
Collaborating to accelerate change
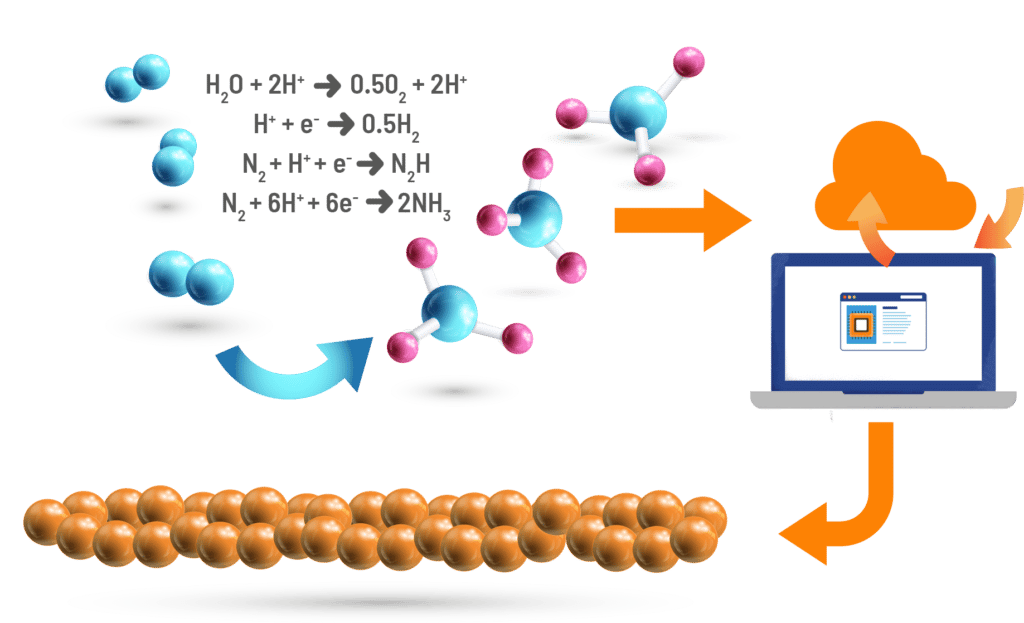
To accelerate the discovery of new electrochemical materials, Citrine has teamed up with research groups from Carnegie Mellon University, MIT, and Julia Computing in a US Department of Energy ARPA-E program called DIFFERENTIATE. The team’s goal is to accelerate electrochemical materials development by 80% through innovating on three key fronts:
- Software and hardware acceleration of high-fidelity simulations that predict catalyst performance
- Fast, accurate surrogate models for time-consuming physics-based simulations
- A Sequential Learning–driven efficient search over a vast chemical design space to identify high-quality candidate materials in as few iterations as possible
The outcomes from this project will advance energy-efficient, sustainable methods of chemical production across a broad range of industries.
Find out more
Details of the technical approach and challenges are discussed in a preprint.